Drug Information
| General Information | ||||||
|---|---|---|---|---|---|---|
| Drug ID |
DR00265
|
|||||
| Drug Name |
Lenvatinib
|
|||||
| Synonyms |
4-[3-chloro-4-(cyclopropylcarbamoylamino)phenoxy]-7-methoxy-quinoline-6-carboxamide; E-7080, E7080; E7080
|
|||||
| Drug Type |
Small molecular drug
|
|||||
| Indication | Ovarian cancer [ICD11: 2C73] | Approved | [1] | |||
| Therapeutic Class |
Anticancer Agents
|
|||||
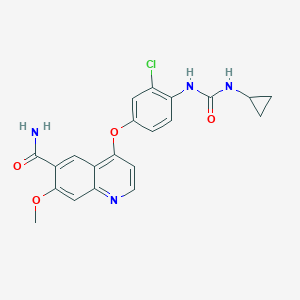
| Structure |
|
 |
||||
| 3D MOL | 2D MOL | |||||
| Formula |
C21H19ClN4O4
|
|||||
| Canonical SMILES |
COC1=CC2=NC=CC(=C2C=C1C(=O)N)OC3=CC(=C(C=C3)NC(=O)NC4CC4)Cl
|
|||||
| InChI |
InChI=1S/C21H19ClN4O4/c1-29-19-10-17-13(9-14(19)20(23)27)18(6-7-24-17)30-12-4-5-16(15(22)8-12)26-21(28)25-11-2-3-11/h4-11H,2-3H2,1H3,(H2,23,27)(H2,25,26,28)
|
|||||
| InChIKey |
WOSKHXYHFSIKNG-UHFFFAOYSA-N
|
|||||
| CAS Number |
CAS 204460-24-2
|
|||||
| Pharmaceutical Properties | Molecular Weight | 426.9 | Topological Polar Surface Area | 116 | ||
| Heavy Atom Count | 30 | Rotatable Bond Count | 6 | |||
| Hydrogen Bond Donor Count | 3 | Hydrogen Bond Acceptor Count | 5 | |||
| XLogP |
2.8
|
|||||
| PubChem CID | ||||||
| PubChem SID |
123098996
, 123110209
, 124757043
, 125163847
, 125749042
, 126665901
, 131314303
, 131480711
, 134221774
, 135262444
, 135626656
, 135685148
, 135685149
, 135685168
, 136367339
, 136367958
, 137262627
, 137276042
, 139802275
, 144115929
, 14782907
, 152258284
, 152344144
, 160647123
, 162011787
, 162037768
, 162527769
, 164041901
, 174560999
, 178103998
, 180386840
, 198978519
, 202553041
, 223669921
, 223705252
, 223913134
, 227134592
, 24126764
, 242060265
, 247802696
, 251911433
, 251971223
, 252150297
, 252215326
, 252215327
, 252451828
, 252543308
, 44859775
, 74753053
, 99436922
|
|||||
| ChEBI ID |
ChEBI:85994
|
|||||
| TTD Drug ID | ||||||
| DT(s) Transporting This Drug | P-GP | Transporter Info | P-glycoprotein 1 | Substrate | [2] | |
| References | ||||||
| 1 | Lenvatinib was approved by FDA. The official website of the U.S. Food and Drug Administration. (2019) | |||||
| 2 | Influence of CYP3A4/5 and ABC transporter polymorphisms on lenvatinib plasma trough concentrations in Japanese patients with thyroid cancer. Sci Rep. 2019 Apr 1;9(1):5404. | |||||
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.
