Drug Information
| General Information | ||||||
|---|---|---|---|---|---|---|
| Drug ID |
DR00600
|
|||||
| Drug Name |
Cefoperazone
|
|||||
| Synonyms |
(6R,7R)-7-((R)-2-(4-Ethyl-2,3-dioxo-1-piperazinylcarboxamido)-2-(4-hydroxyphenyl)acetamido)-3-((1-methyl-1H-tetrazol-5-yl)thiomethyl)-8-oxo-5-thia-1-azabicyclo(4.2.0)oct-2-en-2-carbonsaeure; (6R,7R)-7-[[(2R)-2-[(4-ethyl-2,3-dioxopiperazine-1-carbonyl)amino]-2-(4-hydroxyphenyl)acetyl]amino]-3-[(1-methyltetrazol-5-yl)sulfanylmethyl]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid; (6R,7R)-7-[[(2S)-2-[(4-ethyl-2,3-dioxopiperazine-1-carbonyl)amino]-2-(4-hydroxyphenyl)acetyl]amino]-3-[(1-methyltetrazol-5-yl)sulfanylmethyl]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid; (6R,7R)-7-[[2-[(4-ethyl-2,3-dioxopiperazine-1-carbonyl)amino]-2-(4-hydroxyphenyl)acetyl]amino]-3-[(1-methyltetrazol-5-yl)sulfanylmethyl]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid; (6R,7R)-7-{[(2R)-2-{[(4-ethyl-2,3-dioxopiperazin-1-yl)carbonyl]amino}-2-(4-hydroxyphenyl)acetyl]amino}-3-{[(1-methyl-1H-tetrazol-5-yl)thio]methyl}-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid; (6R,7R)-7-{[{[(4-ethyl-2,3-dioxopiperazin-1-yl)carbonyl]amino}(4-hydroxyphenyl)acetyl]amino}-3-{[(1-methyl-1H-tetrazol-5-yl)sulfanyl]methyl}-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid; (6R,7S)-7-[[(2R)-2-[(4-ethyl-2,3-dioxopiperazine-1-carbonyl)amino]-2-(4-hydroxyphenyl)acetyl]amino]-3-[(1-methyltetrazol-5-yl)sulfanylmethyl]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid; (6R,7S)-7-[[(2S)-2-[(4-ethyl-2,3-dioxopiperazine-1-carbonyl)amino]-2-(4-hydroxyphenyl)acetyl]amino]-3-[(1-methyltetrazol-5-yl)sulfanylmethyl]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid; (6S,7R)-7-[[(2S)-2-[(4-ethyl-2,3-dioxopiperazine-1-carbonyl)amino]-2-(4-hydroxyphenyl)acetyl]amino]-3-[(1-methyltetrazol-5-yl)sulfanylmethyl]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid; 7-[[2-[(4-ethyl-2,3-dioxopiperazine-1-carbonyl)amino]-2-(4-hydroxyphenyl)acetyl]amino]-3-[(1-methyltetrazol-5-yl)sulfanylmethyl]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid; 7beta-{[{[(4-ethyl-2,3-dioxopiperazin-1-yl)carbonyl]amino}(4-hydroxyphenyl)acetyl]amino}-3-[(1-methyl-1H-tetrazol-5-yl)sulfanyl]methyl-3,4-didehydrocepham-4-carboxylic acid; CP 52640; Cefazone (TN); Cefobid; Cefobid (TN); Cefoperazon; Cefoperazone (INN); Cefoperazone [INN:BAN]; Cefoperazone sodium salt; Cefoperazono; Cefoperazono [INN-Spanish]; Cefoperazonum; Cefoperazonum [INN-Latin]; Peracef [veterinary]; Peracef [veterinary] (TN)
|
|||||
| Drug Type |
Small molecular drug
|
|||||
| Indication | Pseudomonas bacterial infections [ICD11: 1B92] | Approved | [1] | |||
| Therapeutic Class |
Antibiotics
|
|||||
| Structure |
|
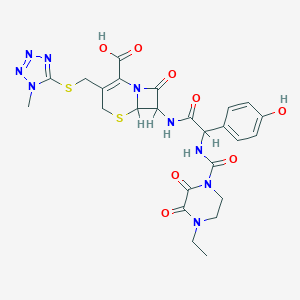 |
||||
| 3D MOL | 2D MOL | |||||
| Formula |
C25H27N9O8S2
|
|||||
| Canonical SMILES |
CCN1CCN(C(=O)C1=O)C(=O)NC(C2=CC=C(C=C2)O)C(=O)NC3C4N(C3=O)C(=C(CS4)CSC5=NN=NN5C)C(=O)O
|
|||||
| InChI |
InChI=1S/C25H27N9O8S2/c1-3-32-8-9-33(21(39)20(32)38)24(42)27-15(12-4-6-14(35)7-5-12)18(36)26-16-19(37)34-17(23(40)41)13(10-43-22(16)34)11-44-25-28-29-30-31(25)2/h4-7,15-16,22,35H,3,8-11H2,1-2H3,(H,26,36)(H,27,42)(H,40,41)
|
|||||
| InChIKey |
GCFBRXLSHGKWDP-UHFFFAOYSA-N
|
|||||
| CAS Number |
CAS 62893-19-0
|
|||||
| Pharmaceutical Properties | Molecular Weight | 645.7 | Topological Polar Surface Area | 271 | ||
| Heavy Atom Count | 44 | Rotatable Bond Count | 9 | |||
| Hydrogen Bond Donor Count | 4 | Hydrogen Bond Acceptor Count | 13 | |||
| XLogP |
-0.7
|
|||||
| PubChem CID | ||||||
| PubChem SID |
104301145
, 11212882
, 11335303
, 11360542
, 11364333
, 11366895
, 11369457
, 11371652
, 11373802
, 11377619
, 11461514
, 11485363
, 11489580
, 11490562
, 11492137
, 11495253
, 125354991
, 125354992
, 144075466
, 163193900
, 175610906
, 179038766
, 223676922
, 242145318
, 244301341
, 251960579
, 29221788
, 47348122
, 47572074
, 47645332
, 50742183
, 5308397
|
|||||
| ChEBI ID |
CHEBI:3493
|
|||||
| TTD Drug ID | ||||||
| DT(s) Transporting This Drug | OATP1B1 | Transporter Info | Organic anion transporting polypeptide 1B1 | Substrate | [2] | |
| OATP1B3 | Transporter Info | Organic anion transporting polypeptide 1B3 | Substrate | [3] | ||
| P-GP | Transporter Info | P-glycoprotein 1 | Substrate | [4] | ||
| References | ||||||
| 1 | Cefoperazone was approved by FDA. The official website of the U.S. Food and Drug Administration. (2019) | |||||
| 2 | Organic anion transporting polypeptide 1B1: a genetically polymorphic transporter of major importance for hepatic drug uptake. Pharmacol Rev. 2011 Mar;63(1):157-81. | |||||
| 3 | Screening of antibiotics that interact with organic anion-transporting polypeptides 1B1 and 1B3 using fluorescent probes. Biol Pharm Bull. 2011;34(3):389-95. | |||||
| 4 | Mammalian drug efflux transporters of the ATP binding cassette (ABC) family in multidrug resistance: A review of the past decade. Cancer Lett. 2016 Jan 1;370(1):153-64. | |||||
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.
