Drug Information
| General Information | ||||||
|---|---|---|---|---|---|---|
| Drug ID |
DR00692
|
|||||
| Drug Name |
Cefdinir
|
|||||
| Synonyms |
(-)-(6R,7R)-7-(2-(2-Amino-4-thiazolyl)glyoxylamido)-8-oxo-3-vinyl-5-thia-1-azabicyclo(4.2.0)oct-2-ene-2-carboxylic acid, 7(sup 2)-(Z)-oxime; (6R,7R)-7-[[(2Z)-2-(2-amino-1,3-thiazol-4-yl)-2-hydroxyiminoacetyl]amino]-3-ethenyl-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid; (6R,7R)-7-{[(2Z)-2-(2-amino-1,3-thiazol-4-yl)-2-(hydroxyimino)acetyl]amino}-3-ethenyl-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid; 7-(2 (2-aminothiazol-4-yl)-2-hydroxyiminoacetamido)-3-vinyl-3-cephem-4-carboxylic acid; 7beta-[(2Z)-2-(2-amino-1,3-thiazol-4-yl)-2-(hydroxyimino)acetamido]-3-ethenyl-3,4-didehydrocepham-4-carboxylic acid; BMY 28488; CFDN; Cefdinir (JP15/USAN/INN); Cefdinir [USAN:INN]; Cefdinirum; Cefdinirum [INN-Latin]; Cefdinyl; Cefdirnir; Cefzon; Cefzon (TN); FK 482; FK-482; FR-80482; KS-1038; Omnicef; Omnicef (TN); Omnicef, FK-482, BMY-28488, PD 134393, CI-983, Cefdinir; PD 134393; PD-134393
|
|||||
| Drug Type |
Small molecular drug
|
|||||
| Indication | Bacterial infections [ICD11: 1A00-1H0Z] | Approved | [1] | |||
| Therapeutic Class |
Antibiotics
|
|||||
| Structure |
|
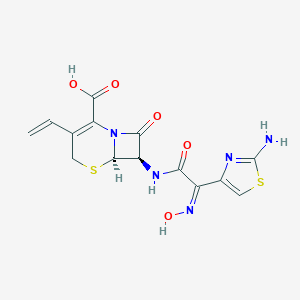 |
||||
| 3D MOL | 2D MOL | |||||
| Formula |
C14H13N5O5S2
|
|||||
| Canonical SMILES |
C=CC1=C(N2C(C(C2=O)NC(=O)C(=NO)C3=CSC(=N3)N)SC1)C(=O)O
|
|||||
| InChI |
InChI=1S/C14H13N5O5S2/c1-2-5-3-25-12-8(11(21)19(12)9(5)13(22)23)17-10(20)7(18-24)6-4-26-14(15)16-6/h2,4,8,12,24H,1,3H2,(H2,15,16)(H,17,20)(H,22,23)/b18-7-/t8-,12-/m1/s1
|
|||||
| InChIKey |
RTXOFQZKPXMALH-GHXIOONMSA-N
|
|||||
| CAS Number |
CAS 91832-40-5
|
|||||
| Pharmaceutical Properties | Molecular Weight | 395.4 | Topological Polar Surface Area | 212 | ||
| Heavy Atom Count | 26 | Rotatable Bond Count | 5 | |||
| Hydrogen Bond Donor Count | 4 | Hydrogen Bond Acceptor Count | 10 | |||
| XLogP |
0
|
|||||
| PubChem CID | ||||||
| PubChem SID |
10310
, 103241607
, 104253245
, 114784936
, 12013492
, 124637292
, 124757367
, 124766167
, 125164171
, 125311797
, 126592899
, 126670303
, 131330280
, 134222573
, 134337732
, 135013391
, 135693788
, 136368001
, 137003422
, 143493270
, 144075787
, 144089123
, 144115805
, 144240157
, 14805465
, 14927923
, 152090681
, 152242898
, 152344175
, 160963880
, 17193404
, 25819898
, 26612828
, 26680572
, 43527984
, 46386833
, 46505573
, 47943747
, 48169352
, 49679095
, 50050916
, 50123150
, 52583972
, 56422579
, 57371624
, 7847980
, 92124803
, 92308050
, 92729947
, 93166985
|
|||||
| ChEBI ID |
CHEBI:3485
|
|||||
| TTD Drug ID | ||||||
| DT(s) Transporting This Drug | OAT3 | Transporter Info | Organic anion transporter 3 | Substrate | [2] | |
| PEPT1 | Transporter Info | Peptide transporter 1 | Substrate | [3] | ||
| References | ||||||
| 1 | Cefdinir was approved by FDA. The official website of the U.S. Food and Drug Administration. (2019) | |||||
| 2 | Human organic anion transporter hOAT3 is a potent transporter of cephalosporin antibiotics, in comparison with hOAT1. Biochem Pharmacol. 2005 Oct 1;70(7):1104-13. | |||||
| 3 | Transporter-mediated drug delivery: recent progress and experimental approaches. Drug Discov Today. 2004 Aug 15;9(16):712-20. | |||||
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.
