Drug Information
| General Information | ||||||
|---|---|---|---|---|---|---|
| Drug ID |
DR00073
|
|||||
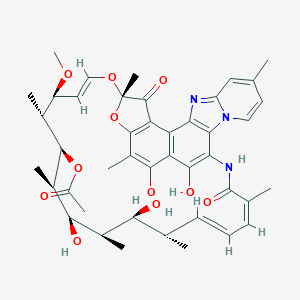
| Drug Name |
Rifaximin
|
|||||
| Synonyms |
Rifaximin; Rifaxidin; Rifacol; Xifaxan; Rifaximine; Rifamycin L 105; Rifamycin L 105SV; Rifaximina; Fatroximin; Normix; Rifaximinum; Rifamixin; Ritacol; Rifaximine [French]; Rifaximinum [Latin]; Rifaximina [Spanish]; Rifaximin [USAN:INN]; UNII-L36O5T016N; L 105SV; L 105 (Ansamacrolide antibiotic); L 105; C43H51N3O11; BRN 3584528; L36O5T016N; Rifaximin, 98%; Rifaximin (Xifaxan); MFCD00864973
|
|||||
| Drug Type |
Small molecular drug
|
|||||
| Indication | Traveler's diarrhea [ICD11: ME05.1] | Approved | [1] | |||
| Irritable bowel syndrome [ICD11: DD91.0] | Approved | [1] | ||||
| Structure |
|
 |
||||
| 3D MOL | 2D MOL | |||||
| Formula |
C43H51N3O11
|
|||||
| Canonical SMILES |
CC1C=CC=C(C(=O)NC2=C(C3=C(C4=C(C(=C3O)C)OC(C4=O)(OC=CC(C(C(C(C(C(C1O)C)O)C)OC(=O)C)C)OC)C)C5=C2N6C=CC(=CC6=N5)C)O)C
|
|||||
| InChI |
InChI=1S/C43H51N3O11/c1-19-14-16-46-28(18-19)44-32-29-30-37(50)25(7)40-31(29)41(52)43(9,57-40)55-17-15-27(54-10)22(4)39(56-26(8)47)24(6)36(49)23(5)35(48)20(2)12-11-13-21(3)42(53)45-33(34(32)46)38(30)51/h11-18,20,22-24,27,35-36,39,48-51H,1-10H3,(H,45,53)/b12-11+,17-15+,21-13-/t20-,22+,23+,24+,27-,35-,36+,39+,43-/m0/s1
|
|||||
| InChIKey |
NZCRJKRKKOLAOJ-XRCRFVBUSA-N
|
|||||
| CAS Number |
CAS 80621-81-4
|
|||||
| Pharmaceutical Properties | Molecular Weight | 785.9 | Topological Polar Surface Area | 198 | ||
| Heavy Atom Count | 57 | Rotatable Bond Count | 3 | |||
| Hydrogen Bond Donor Count | 5 | Hydrogen Bond Acceptor Count | 12 | |||
| XLogP |
6.9
|
|||||
| PubChem CID | ||||||
| ChEBI ID |
CHEBI:75246
|
|||||
| TTD Drug ID | ||||||
| DT(s) Transporting This Drug | P-GP | Transporter Info | P-glycoprotein 1 | Substrate | [2] | |
| References | ||||||
| 1 | Rifaximin was approved by FDA. The official website of the U.S. Food and Drug Administration. (2019) | |||||
| 2 | Advantageous Solubility-Permeability Interplay When Using Amorphous Solid Dispersion (ASD) Formulation for the BCS Class IV P-gp Substrate Rifaximin: Simultaneous Increase of Both the Solubility and the Permeability. AAPS J. 2017 May;19(3):806-813. | |||||
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.
