Drug Information
| General Information | ||||||
|---|---|---|---|---|---|---|
| Drug ID |
DR00299
|
|||||
| Drug Name |
Indinavir
|
|||||
| Synonyms |
(1(1S,2R),5(S))-2,3,5-Trideoxy-N-(2,3-dihydro-2-hydroxy-1H-inden-1-yl)-5-(2-(((1,1-dimethylethyl)amino)carbonyl)-4-(3-pyridinylmethyl)-1-piperazinyl)-2-(phenylmethyl)-D-erythro-Pentonamide; (2S)-1-[(2S,4S)-4-benzyl-2-hydroxy-5-[[(1S,2R)-2-hydroxy-2,3-dihydro-1H-inden-1-yl]amino]-5-oxopentyl]-N-tert-butyl-4-(pyridin-3-ylmethyl)piperazine-2-carboxamide; Crixivan; Crixivan (TM); Crixivan (TN); IDV; Indinavir (*1:1 Sulfate salt*); Indinavir [USAN]; Indinavir, Sulfate (1:1); L 735524; L-735 524; L-735,524; L-735524; MK-639; N-[2(R)-HYDROXY-1(S)-INDANYL]-5-[(2(S)-TERTIARY BUTYLAMINOCARBONYL)-4(3-PYRIDYLMETHYL)PIPERAZINO]-4(S)-HYDROXY-2(R)-PHENYLMETHYLPENTANAMIDE; Propolis & 4-Hydroxy-N-(2-hydroxy-2,3-dihydro-1H-1-indanyl)-N'-(1,1-dimethylethyl)-2-phenylmethyl-5-[4-(3-pyridylmethyl)-1-piperzinyl]hexanediamide; Propolis+Indinavir; RS-253
|
|||||
| Drug Type |
Small molecular drug
|
|||||
| Indication | Human immunodeficiency virus infection [ICD11: 1C62.Z] | Approved | [1] | |||
| Therapeutic Class |
Anti-HIV Agents
|
|||||
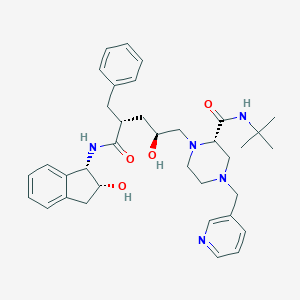
| Structure |
|
 |
||||
| 3D MOL | 2D MOL | |||||
| Formula |
C36H47N5O4
|
|||||
| Canonical SMILES |
CC(C)(C)NC(=O)C1CN(CCN1CC(CC(CC2=CC=CC=C2)C(=O)NC3C(CC4=CC=CC=C34)O)O)CC5=CN=CC=C5
|
|||||
| InChI |
InChI=1S/C36H47N5O4/c1-36(2,3)39-35(45)31-24-40(22-26-12-9-15-37-21-26)16-17-41(31)23-29(42)19-28(18-25-10-5-4-6-11-25)34(44)38-33-30-14-8-7-13-27(30)20-32(33)43/h4-15,21,28-29,31-33,42-43H,16-20,22-24H2,1-3H3,(H,38,44)(H,39,45)/t28-,29+,31+,32-,33+/m1/s1
|
|||||
| InChIKey |
CBVCZFGXHXORBI-PXQQMZJSSA-N
|
|||||
| CAS Number |
CAS 150378-17-9
|
|||||
| Pharmaceutical Properties | Molecular Weight | 613.8 | Topological Polar Surface Area | 118 | ||
| Heavy Atom Count | 45 | Rotatable Bond Count | 12 | |||
| Hydrogen Bond Donor Count | 4 | Hydrogen Bond Acceptor Count | 7 | |||
| XLogP |
2.8
|
|||||
| PubChem CID | ||||||
| PubChem SID |
10317595
, 10317604
, 10317607
, 103179607
, 104322011
, 104829412
, 124892212
, 126628679
, 127336447
, 127336448
, 127336449
, 127336450
, 127602164
, 14765204
, 14862973
, 17422095
, 26697187
, 29211926
, 46391630
, 46392153
, 46392154
, 46392339
, 46393133
, 46393134
, 46393135
, 46393136
, 46393839
, 46393841
, 46393842
, 46393914
, 46506442
, 50064484
, 583813
, 601472
, 7846196
, 7888933
, 7979603
, 8030462
, 8187130
, 822078
, 824057
, 830610
, 832455
, 832456
, 832457
, 834483
, 9263
, 92717923
, 93166536
, 93167159
|
|||||
| ChEBI ID |
ChEBI:44032
|
|||||
| TTD Drug ID | ||||||
| DT(s) Transporting This Drug | MRP1 | Transporter Info | Multidrug resistance-associated protein 1 | Substrate | [2] | |
| MRP2 | Transporter Info | Multidrug resistance-associated protein 2 | Substrate | [3] | ||
| P-GP | Transporter Info | P-glycoprotein 1 | Substrate | [4] | ||
| Drug-Transporter Activity Data | ||||||
| Drug-Transporter Activity Data | P-GP | Transporter Info | Km = 0.47 microM | High five cells-MDR1 | [4] | |
| References | ||||||
| 1 | Indinavir was approved by FDA. The official website of the U.S. Food and Drug Administration. (2019) | |||||
| 2 | Human intestinal transporter database: QSAR modeling and virtual profiling of drug uptake, efflux and interactions. Pharm Res. 2013 Apr;30(4):996-1007. | |||||
| 3 | Interaction of eight HIV protease inhibitors with the canalicular efflux transporter ABCC2 (MRP2) in sandwich-cultured rat and human hepatocytes. Biopharm Drug Dispos. 2010 Mar;31(2-3):178-88. | |||||
| 4 | HIV-1 protease inhibitors are substrates for the MDR1 multidrug transporter. Biochemistry. 1998 Mar 17;37(11):3594-601. | |||||
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.
