Drug Information
| General Information | ||||||
|---|---|---|---|---|---|---|
| Drug ID |
DR00482
|
|||||
| Drug Name |
Irbesartan
|
|||||
| Synonyms |
2-Butyl-3-(p-(o-1H-tetrazol-5-ylphenyl)benzyl)-1,3-diazaspiro(4.4)non-1-en-4-one; 2-Butyl-3-[2'-(1H-tetrazol-5-yl)biphenyl-4-ylmethyl]1,3-diaza-spiro[4.4]non-1-en-4-one; 2-butyl-3-[ p-(o-1 H-tetrazol-5-ylphenyl)benzyl]-1,3-diazaspiro[4,4]non-1-en-4-one; 2-butyl-3-[p-(o-1H-tetrazol-5-ylphenyl)benzyl]-1,3-diazaspiro[4.4]non-1-en-4-one; 2-butyl-3-{[2'-(1H-tetrazol-5-yl)[1,1'-biphenyl]-4-yl]methyl}-1,3-diazaspiro[4.4]non-1-en-4-one; 2-butyl-3-{[2'-(1H-tetrazol-5-yl)biphenyl-4-yl]methyl}-1,3-diazaspiro[4.4]non-1-en-4-one; 2-n-butyl-3-((2'-(1H-tetrazol-5-yl)biphenyl-4-yl)methyl)-1,3-diazaspiro(4,4)non-1-en-4-one; 8-butyl-7-[[4-[2-(2H-tetrazol-5-yl)phenyl]phenyl]methyl]-7,9-diazaspiro[4.4]non-8-en-6-one; Aprovel; Aprovel (TN); Avalide (TN); Avapro; Avapro (TN); BMS 186295; BMS Brand of Irbesartan; BMS-186295; BMS-186295, SR-47436,Aprovel, Karvea, Irbesartan; Bristol Myers Brand of Irbesartan; Irbesarran; Irbesartan (JAN/USAN/INN); Irbesartan [USAN:INN]; Irbetan; Karvea; Karvea (TN); Lrbesartan; SR 47436; SR-47436; SanofiWinthrop Brand of Irbesartan
|
|||||
| Drug Type |
Small molecular drug
|
|||||
| Indication | High blood pressure [ICD11: BA00] | Approved | [1] | |||
| Therapeutic Class |
Antihypertensive Agents
|
|||||
| Structure |
|
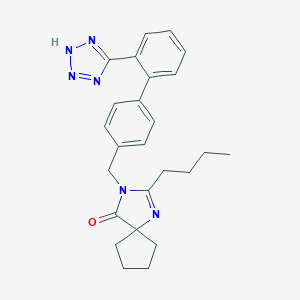 |
||||
| 3D MOL | 2D MOL | |||||
| Formula |
C25H28N6O
|
|||||
| Canonical SMILES |
CCCCC1=NC2(CCCC2)C(=O)N1CC3=CC=C(C=C3)C4=CC=CC=C4C5=NNN=N5
|
|||||
| InChI |
InChI=1S/C25H28N6O/c1-2-3-10-22-26-25(15-6-7-16-25)24(32)31(22)17-18-11-13-19(14-12-18)20-8-4-5-9-21(20)23-27-29-30-28-23/h4-5,8-9,11-14H,2-3,6-7,10,15-17H2,1H3,(H,27,28,29,30)
|
|||||
| InChIKey |
YOSHYTLCDANDAN-UHFFFAOYSA-N
|
|||||
| CAS Number |
CAS 138402-11-6
|
|||||
| Pharmaceutical Properties | Molecular Weight | 428.5 | Topological Polar Surface Area | 87.1 | ||
| Heavy Atom Count | 32 | Rotatable Bond Count | 7 | |||
| Hydrogen Bond Donor Count | 1 | Hydrogen Bond Acceptor Count | 5 | |||
| XLogP |
4.1
|
|||||
| PubChem CID | ||||||
| PubChem SID |
11364645
, 11367207
, 11369769
, 11372006
, 11374741
, 11377931
, 11485625
, 11489491
, 11490809
, 11492936
, 11495565
, 11528645
, 12014641
, 14758693
, 14905264
, 26612692
, 26680708
, 26719813
, 26748950
, 26748951
, 29222869
, 46386566
, 46506575
, 46530573
, 47365300
, 48035232
, 48416128
, 49681744
, 49830881
, 50107493
, 5243560
, 53787775
, 56312044
, 56313979
, 57321974
, 77118987
, 7847589
, 81092816
, 8152372
, 85174437
, 85209120
, 85789259
, 91011661
, 92124749
, 92307926
, 92308458
, 92713301
, 93166505
, 93621098
, 9672
|
|||||
| ChEBI ID |
ChEBI:5959
|
|||||
| TTD Drug ID | ||||||
| DT(s) Transporting This Drug | PEPT1 | Transporter Info | Peptide transporter 1 | Substrate | [2] | |
| References | ||||||
| 1 | Irbesartan was approved by FDA. The official website of the U.S. Food and Drug Administration. (2019) | |||||
| 2 | High-affinity interaction of sartans with H+/peptide transporters. Drug Metab Dispos. 2009 Jan;37(1):143-9. | |||||
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.
