Drug Information
| General Information | ||||||
|---|---|---|---|---|---|---|
| Drug ID |
DR01370
|
|||||
| Drug Name |
Midostaurin
|
|||||
| Synonyms |
PKC412; 120685-11-2; Cgp 41251; 4'-N-Benzoylstaurosporine; CGP-41251; Benzoylstaurosporine; PKC-412; RYDAPT; PKC 412; UNII-ID912S5VON; N-Benzoylstaurosporine; ID912S5VON; CHEMBL608533; CHEBI:63452; Cgp 41 251; N-[(5S,6R,7R,9R)-6-methoxy-5-methyl-14-oxo-6,7,8,9,15,16-hexahydro-5H,14H-5,9-epoxy-4b,9a,15-triazadibenzo[b,h]cyclonona[1,2,3,4-jkl]cyclopenta[e]-as-indacen-7-yl]-N-methylbenzamide; PKC-412(Midostaurin); Midostaurin (PKC412); Midostaurin (USAN/INN); Midostaurin [USAN:INN]; CGP 41231; Rydapt (TN); CPG 41251
|
|||||
| Drug Type |
Small molecular drug
|
|||||
| Indication | Acute myeloid leukemia [ICD11: 2A60] | Approved | [1] | |||
| Therapeutic Class |
Anticancer Agents
|
|||||
| Structure |
|
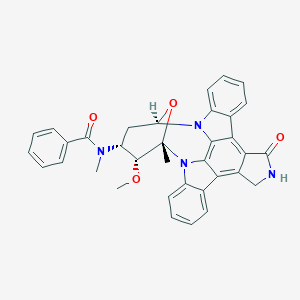 |
||||
| 3D MOL | 2D MOL | |||||
| Formula |
C35H30N4O4
|
|||||
| Canonical SMILES |
CC12C(C(CC(O1)N3C4=CC=CC=C4C5=C6C(=C7C8=CC=CC=C8N2C7=C53)CNC6=O)N(C)C(=O)C9=CC=CC=C9)OC
|
|||||
| InChI |
InChI=1S/C35H30N4O4/c1-35-32(42-3)25(37(2)34(41)19-11-5-4-6-12-19)17-26(43-35)38-23-15-9-7-13-20(23)28-29-22(18-36-33(29)40)27-21-14-8-10-16-24(21)39(35)31(27)30(28)38/h4-16,25-26,32H,17-18H2,1-3H3,(H,36,40)/t25-,26-,32-,35+/m1/s1
|
|||||
| InChIKey |
BMGQWWVMWDBQGC-IIFHNQTCSA-N
|
|||||
| CAS Number |
CAS 120685-11-2
|
|||||
| Pharmaceutical Properties | Molecular Weight | 570.6 | Topological Polar Surface Area | 77.7 | ||
| Heavy Atom Count | 43 | Rotatable Bond Count | 3 | |||
| Hydrogen Bond Donor Count | 1 | Hydrogen Bond Acceptor Count | 4 | |||
| XLogP |
4.8
|
|||||
| PubChem CID | ||||||
| PubChem SID |
14788708
, 14935507
, 22395186
, 44927646
, 47206756
, 53786846
, 57373453
, 79311635
, 99302777
, 103734272
, 123105168
, 124659175
, 124950161
, 134348392
, 135061643
, 135610396
, 137241200
, 143298037
, 172918683
, 174006478
, 177748492
, 178102329
, 179149698
, 184812273
, 198965438
, 210274665
, 210280297
, 223656331
, 233822082
, 241376205
, 248897758
, 249617730
, 252156932
|
|||||
| ChEBI ID |
CHEBI:63452
|
|||||
| TTD Drug ID | ||||||
| DT(s) Transporting This Drug | P-GP | Transporter Info | P-glycoprotein 1 | Substrate | [2] | |
| References | ||||||
| 1 | Midostaurin was approved by FDA. The official website of the U.S. Food and Drug Administration. (2019) | |||||
| 2 | Human intestinal transporter database: QSAR modeling and virtual profiling of drug uptake, efflux and interactions. Pharm Res. 2013 Apr;30(4):996-1007. | |||||
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.
