Drug Information
| General Information | ||||||
|---|---|---|---|---|---|---|
| Drug ID |
DR01375
|
|||||
| Drug Name |
Neostigmine
|
|||||
| Synonyms |
Eustigmin; Eustigmine; Intrastigmina; Juvastigmin; Neostigmin; Neostigminum; Prostigmin; Prostigmine; Prozerin; Sulfonatooxymethane; Synstigmin; Syntostigmine; Vagostigmin; Vagostigmine; Neostigmine [BAN]; Neostigmine omega; M-Trimethylammoniumphenyldimethylcarbamate; Neostigmine (BAN); Prostigmin (TN); Vagostigmin (TN); [3-(dimethylcarbamoyloxy)phenyl]-trimethylazanium; [3-(dimethylcarbamoyloxy)phenyl]-trimethyl-azanium; [3-(dimethylcarbamoyloxy)phenyl]-trimethyl-azanium bromide; Ammonium, (m-hydroxyphenyl)trimethyl-, dimethylcarbamate (ester); Benzenaminium, 3-(((dimethylamino)carbonyl)oxy)-N,N,N-trimethyl-(9CI); (m-Hydroxyphenyl)trimethylammonium dimethylcarbamate; (m-Hydroxyphenyl)trimethylammonium dimethylcarbamate (ester); 3-Trimethylammoniumphenyl N,N-dimethylcarbamate; 3-[(dimethylcarbamoyl)oxy]-N,N,N-trimethylanilinium; 3-{[(dimethylamino)carbonyl]oxy}-N,N,N-trimethylanilinium
|
|||||
| Drug Type |
Small molecular drug
|
|||||
| Indication | Myasthenia gravis [ICD11: 8C60] | Approved | [1] | |||
| Therapeutic Class |
Parasympathomimetics
|
|||||
| Structure |
|
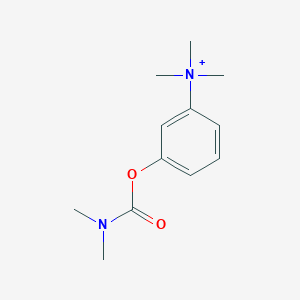 |
||||
| 3D MOL | 2D MOL | |||||
| Formula |
C12H19N2O2+
|
|||||
| Canonical SMILES |
CN(C)C(=O)OC1=CC=CC(=C1)[N+](C)(C)C
|
|||||
| InChI |
InChI=1S/C12H19N2O2/c1-13(2)12(15)16-11-8-6-7-10(9-11)14(3,4)5/h6-9H,1-5H3/q+1
|
|||||
| InChIKey |
ALWKGYPQUAPLQC-UHFFFAOYSA-N
|
|||||
| CAS Number |
CAS 59-99-4
|
|||||
| Pharmaceutical Properties | Molecular Weight | 223.29 | Topological Polar Surface Area | 29.5 | ||
| Heavy Atom Count | 16 | Rotatable Bond Count | 3 | |||
| Hydrogen Bond Donor Count | 0 | Hydrogen Bond Acceptor Count | 2 | |||
| XLogP |
1.5
|
|||||
| PubChem CID | ||||||
| PubChem SID |
9467
, 866380
, 4398035
, 4635954
, 7980106
, 8152746
, 11111527
, 11111528
, 11335340
, 11335373
, 11360579
, 11360612
, 11363955
, 11366517
, 11369079
, 11373762
, 11377241
, 11461551
, 11461584
, 11466496
, 11467616
, 11485012
, 11486251
, 11489094
, 11491887
, 11494875
, 15121488
, 26756597
, 29223551
, 46509161
, 47216650
, 47291007
, 47365046
, 47588860
, 47736334
, 47736335
, 47810618
, 48110324
, 48416319
, 49698982
, 49868416
, 50100289
, 50100290
, 50103950
, 50765645
, 53786926
, 57322279
, 81044623
, 85787484
, 87691541
|
|||||
| ChEBI ID |
CHEBI:7514
|
|||||
| TTD Drug ID | ||||||
| DT(s) Transporting This Drug | P-GP | Transporter Info | P-glycoprotein 1 | Substrate | [2] | |
| References | ||||||
| 1 | Neostigmine was approved by FDA. The official website of the U.S. Food and Drug Administration. (2019) | |||||
| 2 | Improving the prediction of the brain disposition for orally administered drugs using BDDCS. Adv Drug Deliv Rev. 2012 Jan;64(1):95-109. | |||||
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.
