Drug Information
| General Information | ||||||
|---|---|---|---|---|---|---|
| Drug ID |
DR01395
|
|||||
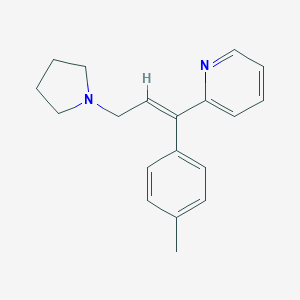
| Drug Name |
Triprolidine
|
|||||
| Synonyms |
Actidil; Actidilat; Entra; Histafed; Myidyl; Tripolidina; Triprolidin; Triprolidinum; Venen; Triprolidine HCL; Triprolidine Monohydrochloride; Triprolidine hydrochloride; Triprolidine hydrochloride anhydrous; T 6764; Actidil (TN); Myidil (TN); Pro-Actidil; Pro-Entra; Tripolidina [INN-Spanish]; Triprolidine (INN); Triprolidine Hydrochloride (anhydrous); Triprolidine Monohydrochloride, Monohydrate; Triprolidine [INN:BAN]; Triprolidinum [INN-Latin]; Venen (TN); Trans-2-(3-(1-Pyrrolidinyl)-1-p-tolylpropenyl)pyridine; Trans-2-(3-(1-Pyrrolidinyl)-1-p-tolylpropenyl)pyridine monohydrochloride; Trans-1-(2-Pyridyl)-3-pyrrolidino-1-p-tolylprop-1-ene; Trans-1-(4-Methylphenyl)-1-(2-pyridyl)-3-pyrrolidinoprop-1-ene; Pyridine, 2-(3-(1-pyrrolidinyl)-1-p-tolylpropenyl)-, monohydrochloride, monohydrate, stereoisomer; Pyridine, 2-((1E)-1-(4-methylphenyl)-3-(1-pyrrolidinyl)-1-propenyl)-, monohydrochloride; Pyridine, 2-((1E)-1-(4-methylphenyl)-3-(1-pyrrolidinyl)-1-propen-1-yl)-, hydrochloride (1:1); (E)-2-(1-(4-Methylphenyl)-3-(1-pyrrolidinyl)-1-propenyl)pyridine; (E)-2-[3-(1-Pyrrolidinyl)-1-p-tolylpropenyl]pyridine hydrochloride; 2-[(E)-1-(4-methylphenyl)-3-pyrrolidin-1-ium-1-ylprop-1-enyl]pyridine chloride; 2-[(E)-1-(4-methylphenyl)-3-pyrrolidin-1-ylprop-1-enyl]pyridine; 2-[(E)-1-(4-methylphenyl)-3-pyrrolidin-1-ylprop-1-enyl]pyridine hydrochloride
|
|||||
| Drug Type |
Small molecular drug
|
|||||
| Indication | Allergic rhinitis [ICD11: CA08.0] | Approved | [1] | |||
| Therapeutic Class |
Antiallergic Agents
|
|||||
| Structure |
|
 |
||||
| 3D MOL | 2D MOL | |||||
| Formula |
C19H22N2
|
|||||
| Canonical SMILES |
CC1=CC=C(C=C1)C(=CCN2CCCC2)C3=CC=CC=N3
|
|||||
| InChI |
InChI=1S/C19H22N2/c1-16-7-9-17(10-8-16)18(19-6-2-3-12-20-19)11-15-21-13-4-5-14-21/h2-3,6-12H,4-5,13-15H2,1H3/b18-11+
|
|||||
| InChIKey |
CBEQULMOCCWAQT-WOJGMQOQSA-N
|
|||||
| CAS Number |
CAS 486-12-4
|
|||||
| Pharmaceutical Properties | Molecular Weight | 278.4 | Topological Polar Surface Area | 16.1 | ||
| Heavy Atom Count | 21 | Rotatable Bond Count | 4 | |||
| Hydrogen Bond Donor Count | 0 | Hydrogen Bond Acceptor Count | 2 | |||
| XLogP |
3.9
|
|||||
| PubChem CID | ||||||
| PubChem SID |
7980840
, 8077587
, 11038508
, 14897473
, 17448291
, 26751829
, 39315888
, 46505403
, 47291040
, 47440150
, 47736373
, 47736374
, 47885310
, 47885311
, 48110356
, 48334387
, 49698410
, 49855442
, 50104523
, 50104524
, 50104525
, 57309729
, 75497893
, 85209605
, 85788432
, 90341007
, 92308937
, 93166727
, 96025331
, 103223987
, 104133962
, 113857221
, 124610340
, 124886935
, 124886936
, 134338323
, 134975511
, 135651281
, 137002512
, 138314072
, 140229550
, 144204470
, 160963773
, 170464931
, 172820007
, 175267951
, 176483998
, 179113821
, 181753769
, 181855806
|
|||||
| ChEBI ID |
CHEBI:84116
|
|||||
| TTD Drug ID | ||||||
| DT(s) Transporting This Drug | P-GP | Transporter Info | P-glycoprotein 1 | Substrate | [2] | |
| References | ||||||
| 1 | Triprolidine was approved by FDA. The official website of the U.S. Food and Drug Administration. (2019) | |||||
| 2 | Central nervous system pharmacokinetics of the Mdr1 P-glycoprotein substrate CP-615,003: intersite differences and implications for human receptor occupancy projections from cerebrospinal fluid exposures. Drug Metab Dispos. 2007 Aug;35(8):1341-9. | |||||
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.
