Drug Information
| General Information | ||||||
|---|---|---|---|---|---|---|
| Drug ID |
DR00206
|
|||||
| Drug Name |
Pitavastatin calcium
|
|||||
| Synonyms |
147526-32-7; 2C25H23FNO4Ca; AK-50694; Alipza; Bis((3R,5S,6E)-7-(2-cyclopropyl-4-(4-fluorophenyl)-3-quinolyl)-3,5-dihydroxy-6-heptenoate), monocalcium salt; CHEBI:71258; Calcium (3R,5S,E)-7-(2-cyclopropyl-4-(4-fluorophenyl)quinolin-3-yl)-3,5-dihydroxyhept-6-enoate; Flovas; IYD54XEG3W; Itavastatin calcium; Livalo; Livazo; NK 104 (acid); NK-104; Nisvastatin; Pitavastatin calcium; Pitavastatin calcium (JAN); Pitavastatin hemicalcium; UNII-IYD54XEG3W
|
|||||
| Drug Type |
Small molecular drug
|
|||||
| Indication | Dyslipidaemias [ICD11: 5C8Z] | Approved | [1] | |||
| Structure |
|
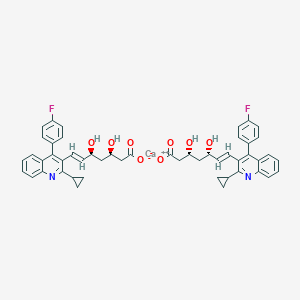 |
||||
| 3D MOL | 2D MOL | |||||
| Formula |
C50H46CaF2N2O8
|
|||||
| Canonical SMILES |
C1CC1C2=NC3=CC=CC=C3C(=C2C=CC(CC(CC(=O)[O-])O)O)C4=CC=C(C=C4)F.C1CC1C2=NC3=CC=CC=C3C(=C2C=CC(CC(CC(=O)[O-])O)O)C4=CC=C(C=C4)F.[Ca+2]
|
|||||
| InChI |
InChI=1S/2C25H24FNO4.Ca/c2*26-17-9-7-15(8-10-17)24-20-3-1-2-4-22(20)27-25(16-5-6-16)21(24)12-11-18(28)13-19(29)14-23(30)31;/h2*1-4,7-12,16,18-19,28-29H,5-6,13-14H2,(H,30,31);/q;;+2/p-2/b2*12-11+;/t2*18-,19-;/m11./s1
|
|||||
| InChIKey |
RHGYHLPFVJEAOC-FFNUKLMVSA-L
|
|||||
| CAS Number |
CAS 147526-32-7
|
|||||
| Pharmaceutical Properties | Molecular Weight | 881 | Topological Polar Surface Area | 187 | ||
| Heavy Atom Count | 63 | Rotatable Bond Count | 14 | |||
| Hydrogen Bond Donor Count | 4 | Hydrogen Bond Acceptor Count | 12 | |||
| PubChem CID | ||||||
| ChEBI ID |
CHEBI:71258
|
|||||
| TTD Drug ID | ||||||
| DT(s) Transporting This Drug | BCRP | Transporter Info | Breast cancer resistance protein | Substrate | [2] | |
| MRP2 | Transporter Info | Multidrug resistance-associated protein 2 | Substrate | [3] | ||
| NTCP | Transporter Info | Sodium/taurocholate cotransporting polypeptide | Substrate | [4] | ||
| OAT3 | Transporter Info | Organic anion transporter 3 | Substrate | [4] | ||
| OATP1A2 | Transporter Info | Organic anion transporting polypeptide 1A2 | Substrate | [4] | ||
| OATP1B1 | Transporter Info | Organic anion transporting polypeptide 1B1 | Substrate | [5] | ||
| OATP1B3 | Transporter Info | Organic anion transporting polypeptide 1B3 | Substrate | [6] | ||
| OATP2B1 | Transporter Info | Organic anion transporting polypeptide 2B1 | Substrate | [7] | ||
| Drug-Transporter Activity Data | ||||||
| Drug-Transporter Activity Data | BCRP | Transporter Info | Km = 5.73 microM | Human embryonic kidney cells (HEK293)-BCRP | [2] | |
| NTCP | Transporter Info | Km = 15.1 microM | Oocytes-NTCP | [4] | ||
| OAT3 | Transporter Info | Km = 3.3 microM | Oocytes-OAT3 | [4] | ||
| OATP1A2 | Transporter Info | Km = 3.4 microM | Oocytes-OATP1A2 | [4] | ||
| OATP1B1 | Transporter Info | Km = 3 microM | Human embryonic kidney cells (HEK293)-OATP1B1 | [6] | ||
| OATP1B1 | Transporter Info | Km = 4.8 microM | Human embryonic kidney cells (HEK293)-OATP1B1 | [8] | ||
| OATP1B1 | Transporter Info | Km = 3.6 microM | Oocytes-OATP1B1 | [4] | ||
| OATP1B1 | Transporter Info | Km = 6.7 microM | Oocytes-OATP1B1 | [5] | ||
| OATP1B3 | Transporter Info | Km = 3.25 microM | Human embryonic kidney cells (HEK293)-OATP1B3 | [6] | ||
| OATP1B3 | Transporter Info | Km = 3.8 microM | Oocytes-OATP1B3 | [4] | ||
| OATP2B1 | Transporter Info | Km = 1.17 microM | Human embryonic kidney cells (HEK293)-OATP2B1 | [7] | ||
| References | ||||||
| 1 | Pitavastatin was approved by FDA. The official website of the U.S. Food and Drug Administration. (2019) | |||||
| 2 | Involvement of BCRP (ABCG2) in the biliary excretion of pitavastatin. Mol Pharmacol. 2005 Sep;68(3):800-7. | |||||
| 3 | Mammalian drug efflux transporters of the ATP binding cassette (ABC) family in multidrug resistance: A review of the past decade. Cancer Lett. 2016 Jan 1;370(1):153-64. | |||||
| 4 | Transporter-mediated influx and efflux mechanisms of pitavastatin, a new inhibitor of HMG-CoA reductase. J Pharm Pharmacol. 2005 Oct;57(10):1305-11. | |||||
| 5 | The effect of SLCO1B1*15 on the disposition of pravastatin and pitavastatin is substrate dependent: the contribution of transporting activity changes by SLCO1B1*15. Pharmacogenet Genomics. 2008 May;18(5):424-33. | |||||
| 6 | Contribution of OATP2 (OATP1B1) and OATP8 (OATP1B3) to the hepatic uptake of pitavastatin in humans. J Pharmacol Exp Ther. 2004 Oct;311(1):139-46. | |||||
| 7 | Drug-drug interaction between pitavastatin and various drugs via OATP1B1. Drug Metab Dispos. 2006 Jul;34(7):1229-36. | |||||
| 8 | The development, characterization, and application of an OATP1B1 inhibition assay in drug discovery. Drug Metab Dispos. 2012 Aug;40(8):1641-8. | |||||
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.
