Drug Information
| General Information | ||||||
|---|---|---|---|---|---|---|
| Drug ID |
DR00588
|
|||||
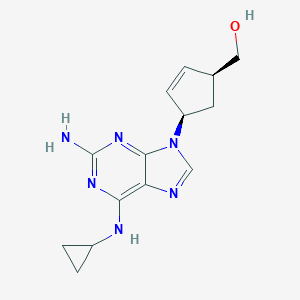
| Drug Name |
Abacavir
|
|||||
| Synonyms |
(+/-)-4-[2-Amino-6-(cyclopropylamino)-9H-purin-9-yl]-2-cyclopentene-1-methanol; (+/-)-Abacavir; (1S,4R)-4-[2-Amino-6-(cyclopropylamino)-9H-purin-9-yl]-2-cyclopentene-1-methanol; ABC; Abacavir (INN); Abacavir [INN]; Trizivir; Ziagen; Ziagen (TM)(*Succinate salt*); Ziagen (TN); [(1S,4R)-4-[2-amino-6-(cyclopropylamino)purin-9-yl]cyclopent-2-en-1-yl]methanol; {(1S-cis)-4-[2-amino-6-(cyclopropylamino)-9H-purin-9-yl]cyclopent-2-en-1-yl}methanol
|
|||||
| Drug Type |
Small molecular drug
|
|||||
| Indication | Human immunodeficiency virus infection [ICD11: 1C62.Z] | Approved | [1] | |||
| Therapeutic Class |
Anti-HIV Agents
|
|||||
| Structure |
|
 |
||||
| 3D MOL | 2D MOL | |||||
| Formula |
C14H18N6O
|
|||||
| Canonical SMILES |
C1CC1NC2=C3C(=NC(=N2)N)N(C=N3)C4CC(C=C4)CO
|
|||||
| InChI |
InChI=1S/C14H18N6O/c15-14-18-12(17-9-2-3-9)11-13(19-14)20(7-16-11)10-4-1-8(5-10)6-21/h1,4,7-10,21H,2-3,5-6H2,(H3,15,17,18,19)/t8-,10+/m1/s1
|
|||||
| InChIKey |
MCGSCOLBFJQGHM-SCZZXKLOSA-N
|
|||||
| CAS Number |
CAS 136470-78-5
|
|||||
| Pharmaceutical Properties | Molecular Weight | 286.33 | Topological Polar Surface Area | 102 | ||
| Heavy Atom Count | 21 | Rotatable Bond Count | 4 | |||
| Hydrogen Bond Donor Count | 3 | Hydrogen Bond Acceptor Count | 6 | |||
| XLogP |
0.9
|
|||||
| PubChem CID | ||||||
| PubChem SID |
10298786
, 103463308
, 104052305
, 104625144
, 126654052
, 128575040
, 131298145
, 135025044
, 136345668
, 137006087
, 142178555
, 14775540
, 14799909
, 160845918
, 160964383
, 162011475
, 162176802
, 163621096
, 163686421
, 163883088
, 164814948
, 165235811
, 175267614
, 175442679
, 179149865
, 184545024
, 186014779
, 196109919
, 223516662
, 223703089
, 223704757
, 223900945
, 226424139
, 241033176
, 241383802
, 26757979
, 26757980
, 36885172
, 46505718
, 51091399
, 57403623
, 603460
, 612310
, 70504220
, 76364492
, 7978623
, 81092778
, 85612588
, 92729708
, 9826
|
|||||
| ChEBI ID |
ChEBI:2360
|
|||||
| TTD Drug ID | ||||||
| DT(s) Transporting This Drug | BCRP | Transporter Info | Breast cancer resistance protein | Substrate | [2] | |
| MRP4 | Transporter Info | Multidrug resistance-associated protein 4 | Substrate | [3] | ||
| P-GP | Transporter Info | P-glycoprotein 1 | Substrate | [4] | ||
| References | ||||||
| 1 | Cidofovir was approved by FDA. The official website of the U.S. Food and Drug Administration. (2019) | |||||
| 2 | QSAR analysis and molecular modeling of ABCG2-specific inhibitors. Adv Drug Deliv Rev. 2009 Jan 31;61(1):34-46. | |||||
| 3 | Human intestinal transporter database: QSAR modeling and virtual profiling of drug uptake, efflux and interactions. Pharm Res. 2013 Apr;30(4):996-1007. | |||||
| 4 | The transport of anti-HIV drugs across blood-CNS interfaces: summary of current knowledge and recommendations for further research. Antiviral Res. 2009 May;82(2):A99-109. | |||||
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.
