Drug Information
| General Information | ||||||
|---|---|---|---|---|---|---|
| Drug ID |
DR01277
|
|||||
| Drug Name |
Entecavir
|
|||||
| Synonyms |
2-amino-1,9-dihydro-9-[(1S,3R,4S)-4-hydroxy-3-(hydroxymethyl)-2-methylenecyclopentyl]-6H-purin-6-one; 2-amino-9-[(1S,3R,4S)-4-hydroxy-3-(hydroxymethyl)-2-methylidenecyclopentyl]-1,9-dihydro-6H-purin-6-one; 2-amino-9-[(1S,3R,4S)-4-hydroxy-3-(hydroxymethyl)-2-methylidenecyclopentyl]-1,9-dihydro-6H-purin-6-one-water (1/1); 2-amino-9-[(1S,3R,4S)-4-hydroxy-3-(hydroxymethyl)-2-methylidenecyclopentyl]-3H-purin-6-one; 6-H-Purin-6-one-,2-amino-1,9-dihydro-9-[(1S,3R,4S)-4-hydroxy-3-(hydroxymethyl)-2-methylenecyclopentyl]; 9-((1S,3R,4S)-4-hydroxy-3-(hydroxymethyl)-2-methylenecyclopentyl)guanine monohydrate; BMS-200475; Baraclude; Baraclude (TN); ETV; Entecavir (INN); Entecavir (USAN); Entecavir hydrate; Entecavir hydrate (JAN); Entecavir monohydrate; SQ-34676
|
|||||
| Drug Type |
Small molecular drug
|
|||||
| Indication | Chronic hepatitis B infection [ICD11: 1E51.0] | Approved | [1] | |||
| Therapeutic Class |
Antiviral Agents
|
|||||
| Structure |
|
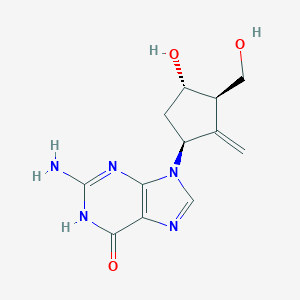 |
||||
| 3D MOL | 2D MOL | |||||
| Formula |
C12H15N5O3
|
|||||
| Canonical SMILES |
C=C1C(CC(C1CO)O)N2C=NC3=C2N=C(NC3=O)N
|
|||||
| InChI |
InChI=1S/C12H15N5O3/c1-5-6(3-18)8(19)2-7(5)17-4-14-9-10(17)15-12(13)16-11(9)20/h4,6-8,18-19H,1-3H2,(H3,13,15,16,20)/t6-,7-,8-/m0/s1
|
|||||
| InChIKey |
QDGZDCVAUDNJFG-FXQIFTODSA-N
|
|||||
| CAS Number |
CAS 142217-69-4
|
|||||
| Pharmaceutical Properties | Molecular Weight | 277.28 | Topological Polar Surface Area | 126 | ||
| Heavy Atom Count | 20 | Rotatable Bond Count | 2 | |||
| Hydrogen Bond Donor Count | 4 | Hydrogen Bond Acceptor Count | 5 | |||
| XLogP |
-1.3
|
|||||
| PubChem CID | ||||||
| PubChem SID |
17398002
, 37101953
, 49830936
, 56391942
, 57379683
, 91749140
, 99319526
, 104253461
, 118313779
, 124757591
, 125001900
, 125164395
, 126651626
, 134222146
, 136920321
, 137129051
, 137179356
, 152034465
, 162011500
, 162183301
, 163370951
, 163908003
, 164825059
, 164833113
, 172087090
, 172440023
, 174531851
, 175265673
, 180672743
, 186013131
, 196106268
, 198991663
, 210274904
, 210280539
, 223379249
, 223398838
, 226415922
, 249739265
, 251893489
, 252110220
, 252162333
, 252316076
|
|||||
| ChEBI ID |
CHEBI:473990
|
|||||
| TTD Drug ID | ||||||
| DT(s) Transporting This Drug | BCRP | Transporter Info | Breast cancer resistance protein | Substrate | [2] | |
| CNT2 | Transporter Info | Concentrative nucleoside transporter 2 | Substrate | [2] | ||
| CNT3 | Transporter Info | Concentrative Na(+)-nucleoside cotransporter 3 | Substrate | [2] | ||
| ENT1 | Transporter Info | Equilibrative nucleoside transporter 1 | Substrate | [2] | ||
| ENT2 | Transporter Info | Equilibrative nucleoside transporter 2 | Substrate | [2] | ||
| OAT2 | Transporter Info | Organic anion transporter 2 | Substrate | [3] | ||
| OCT-3 | Transporter Info | Organic cation transporter 3 | Substrate | [2] | ||
| OCTN2 | Transporter Info | Organic cation/carnitine transporter 2 | Substrate | [2] | ||
| PEPT2 | Transporter Info | Peptide transporter 2 | Substrate | [4] | ||
| Drug-Transporter Activity Data | ||||||
| Drug-Transporter Activity Data | CNT2 | Transporter Info | Km = 53.2 microM | Madin-Darby canine kidney (MDCK) cells-hCNT2 | [2] | |
| CNT3 | Transporter Info | Km = 23.1 microM | Madin-Darby canine kidney (MDCK) cells-hCNT3 | [2] | ||
| References | ||||||
| 1 | Entecavir was approved by FDA. The official website of the U.S. Food and Drug Administration. (2019) | |||||
| 2 | Multiple SLC and ABC Transporters Contribute to the Placental Transfer of Entecavir. Drug Metab Dispos. 2017 Mar;45(3):269-278. | |||||
| 3 | Human organic anion transporter 2 is an entecavir, but not tenofovir, transporter. Drug Metab Pharmacokinet. 2017 Feb;32(1):116-119. | |||||
| 4 | The oligopeptide transporter 2-mediated reabsorption of entecavir in rat kidney. Eur J Pharm Sci. 2014 Feb 14;52:41-7. | |||||
If you find any error in data or bug in web service, please kindly report it to Dr. Yin and Dr. Li.
